A Conclusive Help guide ParaGard Lawsuits
Categories:
ParaGard is among many intrauterine health-related devices (IUDs) in the marketplace that are inserted in ladies as a birth control product. Like several IUDs, ParaGard is meant to be an easily easily-removed, temporary contraceptive – ParaGard is now accepted to be utilized continually for as much as ten years. Given that its preliminary acceptance by america Food items and Medication Supervision in 1984, ParaGard continues to be produced by a variety of firms, including Cooper, GynoPharma, GynoMed, Ortho-McNeil, FEI Women’s Overall health, Duramed Pharmaceuticals, Barr Pharmaceuticals, and Teva Pharmaceutic Market sectors.
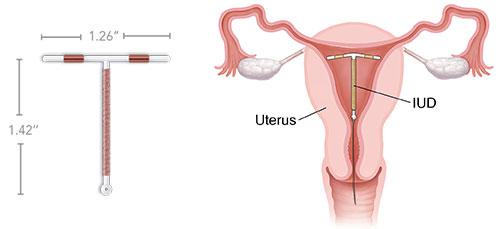
ParaGard removal side effects is different from various other IUDs in that we now have no medications or chemicals involved. Alternatively, there is certainly a small amount of copper in ParaGard that communicates with a woman’s entire body to make a contraceptive chemical substance reaction. That copper is coiled around a compact piece of plastic shaped like the note T. The entire system is below 1.5 by 1.5 inches wide and weighs less than a gram. The Paragard IUD is nonsurgically placed in the uterus by using a specialised system. Threads linked to the Paragard IUD are later useful for nonsurgical elimination of these devices.
However, some types of ParaGard (in particular the 380 designs) have shown to have brittle hands that are given to breakage, particularly when doctors try and eliminate the device. Once these components of the ParaGard IUD crack away from, they can migrate during the entire body and lead to serious injuries. Appropriately, a huge selection of women across the nation have introduced lawsuits for injuries continual as a result of ParaGard IUD damage and ensuing injuries.
All those law suits allege how the companies of ParaGard understand that these IUDs are flawed and therefore patients were improperly warned of the hazards of ParaGard IUDs. Despite, these problems have not even triggered a recall in the very lucrative ParaGard T 380A IUDs, which stay available today.


